Executive Summary
Bakuchiol may be positioned as a gentler retinol alternative, but real-world performance is often decided by the delivery system— not the headline ingredient. Published clinical and formulation research supports bakuchiol’s visible skin-aging benefits and highlights why encapsulation can improve practical outcomes like stability, sensorial acceptance, and consistency in use. This article breaks down the formulation checkpoints that matter most—compatibility, processing, and tolerability—and shows how a delivery-led bakuchiol system can help teams build retinol-alternative formulas that feel better and work better.
Bakuchiol Is Trending—But Delivery System Design Determines Whether It Performs
Retinol alternatives are no longer a niche conversation. They are a formulation brief.
Recent industry coverage makes that clear: brands continue launching retinol-alternative products for consumers who want visible anti-aging results but struggle with the irritation profile often associated with retinoid routines. In that same discussion, bakuchiol remains one of the most visible alternatives, especially in products positioned for sensitive skin, barrier-aware routines, and long-term use tolerance.
That market momentum is important—but it can also create a formulation blind spot. Bakuchiol demand does not automatically translate into bakuchiol performance. For brand and R&D teams, the relevant question is not simply whether bakuchiol is in the formula. It is whether the delivery architecture supports stability, skin compatibility, and real-world use in the format you want to launch.
Why Retinol Alternatives Are Moving from Trend to Formulation Requirement
Global Cosmetic Industry’s January 2026 coverage frames the current market shift well: retinol remains a benchmark ingredient, but consumer experience continues to shape product adoption. Dryness, stinging, peeling, redness, and sensitivity concerns are part of why brands keep expanding alternatives and retinol-adjacent strategies.
That matters for formulators because the brief has changed. Many teams are no longer asked only for “maximum activity.” They are asked for visible improvement with better routine compliance, especially for users who do not tolerate aggressive regimens well. In practice, that means balancing efficacy, sensory feel, barrier support, and irritation management from the first prototype—not trying to fix tolerability after claim direction is already set.
The same article also shows something else: the category is maturing beyond single-ingredient storytelling. Market players are pairing bakuchiol with peptides, barrier-supportive systems, or encapsulated actives, and they are increasingly talking about how the formula is built, not just what the hero active is. That is exactly where a technical marketing bridge becomes valuable.
Why Bakuchiol Keeps Showing Up in Anti-Aging Product Development
Bakuchiol is not “new,” but its current momentum is supported by a more established evidence base than many trend ingredients. A randomized, double-blind clinical comparison cited in PubMed (44 patients, 12 weeks) reported that topical bakuchiol and retinol both significantly reduced wrinkle surface area and hyperpigmentation, with no statistical difference between them in those endpoints, while retinol users reported more scaling and stinging.
That does not mean bakuchiol and retinol are interchangeable in every use case. It does mean bakuchiol has earned a legitimate place in anti-aging formulation planning—especially when tolerability is part of the product promise. This is also consistent with later peer-reviewed discussions describing bakuchiol as a promising anti-aging active with a favorable tolerability profile, while still calling for continued research and careful claim framing.
A second reason bakuchiol remains attractive to brand teams is positioning flexibility. It can fit anti-aging, smoothing, tone-evening, and soothing narratives, depending on the total formulation design and substantiation path. That flexibility is commercially useful—but only if the formulation system preserves the active and supports delivery in the intended product format.
The Real Formulation Challenge: Bakuchiol on the Label vs. Bakuchiol in the Skin
This is where many trend-driven concepts lose momentum during development. A formula can list bakuchiol and still underperform if the active isn’t adequately protected, isn’t dispersed in a way that stays uniform over time, or isn’t presented to the skin consistently across real-world use. The result is familiar: a strong story in the concept deck, but uneven outcomes in stability, sensory, or perceived performance.
In other words, a label claim is not the same thing as functional delivery. Delivery is not a marketing detail; it’s the mechanism that determines whether bakuchiol remains available in the base, survives the environment you’ve built around it, and reaches the skin in a form that can do work.
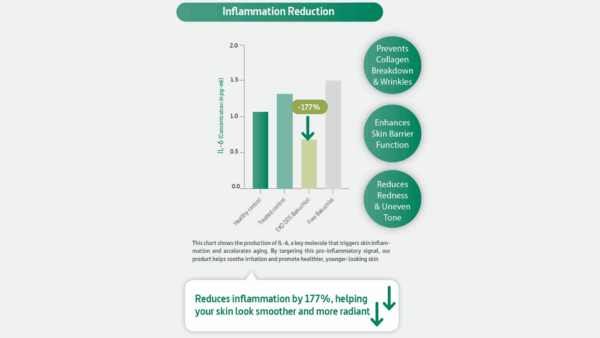
Peer-reviewed formulation work on bakuchiol reinforces why delivery technologies matter. For example, a 2024 pilot study comparing a bakuchiol oil cream with an encapsulated bakuchiol cream reported stronger outcomes for the encapsulated format within the study context, including discussion around hydration, pore visibility, and sebum-related effects versus the non-encapsulated comparator. The authors also point to pragmatic formulation benefits—such as masking color/odor and improving cream stability—which directly affect consumer acceptance and product consistency.
That’s the technical lesson for cosmetic formulators: if you’re building a retinol-alternative product around bakuchiol, delivery system selection is simultaneously an efficacy decision, a stability decision, and a tolerability decision. It’s one lever that influences all three.
This is also why “bakuchiol is trending” can be incomplete from an R&D perspective. Trend coverage is useful for market timing. But formulation success is decided by compatibility, process conditions, phase behavior, surfactant and solvent exposure, packaging strategy, and how the active system behaves over time in the finished base—because those factors determine whether the consumer experiences “gentle and effective,” or simply “present on the label.”
What to Evaluate in a Bakuchiol Delivery System Before You Build Claims
Before locking in messaging, it helps to evaluate a bakuchiol ingredient system through a formulation lens first.
1) Active format and delivery mechanism
Not all bakuchiol inputs behave the same once they hit a real base. A free bakuchiol oil, a solubilized concentrate, and a vesicle/encapsulated system can all support a “bakuchiol” claim, but they won’t deliver the same outcomes in stability, dispersion, or skin feel. Delivery architecture can influence how consistently the active is presented to the skin over time—and whether performance holds up across the shelf life you need.
Peer-reviewed formulation work on bakuchiol supports this point: encapsulation approaches have been reported to improve practical factors like cream stability and sensorial acceptance, and may influence visible outcomes compared with non-encapsulated formats. The takeaway for formulators is simple: choose a bakuchiol format for the formula you’re building, not just the claim you want to make.
2) System compatibility and process constraints
Compatibility is where “good on paper” concepts often stall. Delivery-based systems can be more sensitive to their environment than free oils, so it’s worth treating your base as part of the active system. Solvent load, surfactant intensity, electrolyte content, and mixing energy can all shift how a delivery format behaves—sometimes without obvious instability until you run a real stability program.
For vesicle-based systems specifically, INdermal notes practical constraints that can impact performance: higher alcohol environments and harsher detergent/surfactant conditions may compromise vesicle integrity, and addition strategy matters for batch-to-batch consistency. These considerations are less about restrictions and more about control—keeping processing conditions aligned with how the delivery system is designed to function.
3) Application fit
A strong bakuchiol system should match the usage pattern that supports benefit perception. Leave-on formats typically provide the most consistent consumer experience because contact time is long and routine compliance is easier to maintain. That makes serums and creams common landing zones for retinol-alternative concepts, while masks and ampoules can also make sense when the base is compatible and the delivery format remains stable.
The most useful way to assess “format fit” is to prototype the active system in your intended texture families (watery serum, gel-cream, lotion, richer cream) and compare not only stability and feel, but also how cleanly the concept supports your claim story.
4) Claim substantiation quality
If you plan to use performance data in marketing, the first step is to verify what the data actually represents. The most reliable claim decisions come from understanding the study type (in vitro, ex vivo, in vivo), the comparator, the dose, treatment duration, and whether the endpoint maps cleanly to the claim language you want to use.
INdermal’s documentation for delivery-led actives may include mechanism and performance-oriented data; treat that as a strong starting point, then decide what must be confirmed in your finished formula (especially if you’re changing the base architecture, adding potential destabilizers, or targeting sensitive-skin positioning).
A Delivery-Led Bakuchiol Approach for Better Stability, Tolerability, and Use Flexibility
The practical value of this topic is showing how a delivery-led bakuchiol system can help close the gap between market demand and formulation execution.
EXO DDS Bakuchiol is positioned as a 1% bakuchiol ingredient encapsulated in exosome-like deep delivery nanovesicles with avocado-derived components, with the product description emphasizing support for penetration, stability, prolonged activity, and lower irritation potential relative to conventional approaches.
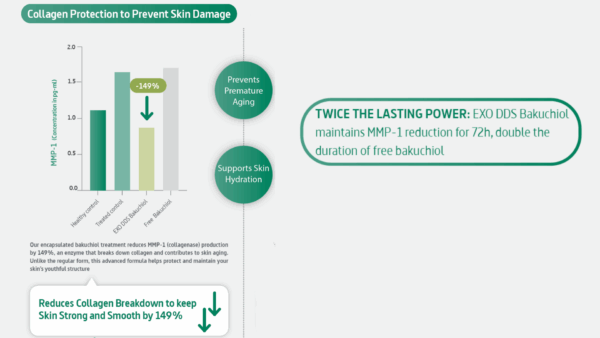
What makes this relevant in a technical marketing context is not just the headline. It is the combination of:
- a ready-to-use delivery format
- explicit formulation handling guidance
- broad application positioning (serums/creams/masks/ampoules)
- a claim direction that aligns with current retinol-alternative briefs (firmness, soothing, rejuvenation, texture support).
That gives both sides of the development team something useful. R&D gets process and compatibility clues that reduce trial-and-error. Brand teams get a more defensible story centered on delivery and tolerability strategy instead of generic “clean anti-aging” language.
It also aligns with the broader trend direction seen in industry coverage, where brands and suppliers increasingly emphasize encapsulation, combination systems, and barrier-aware design rather than relying on one active to carry the entire performance narrative.
How Brand and R&D Teams Can Build Stronger Retinol-Alternative Claims
A better retinol-alternative product story starts by avoiding overreach.
Instead of broad claims that imply equivalence in every function, stronger positioning usually comes from specific, substantiated, and formulation-aware language, such as:
- Supports visible smoothing and firmness in anti-aging skin care routines
- Designed for bakuchiol delivery in aqueous-compatible systems
- Built for retinol-alternative concepts where tolerability is a design priority
- Suitable for serum and cream formats requiring controlled active delivery
This kind of phrasing gives marketing teams room to communicate benefits while staying grounded in what can actually be supported by ingredient documentation and final-formula testing.
Just as importantly, it helps internal alignment. When marketing and R&D use the same technical logic—delivery system, compatibility, tolerability, endpoint relevance—the path from concept sheet to launch copy gets much smoother.
Don’t Just Choose Bakuchiol—Choose the Right Bakuchiol System
Bakuchiol has earned its place in the retinol-alternative conversation. The evidence base and market momentum both support that. But the next level of product performance comes from a more disciplined question:
What delivery system are you choosing, and what does that choice do for stability, tolerability, and formulation robustness?
For formulators and brand developers, that is where the real differentiation begins—especially in a crowded category where many products make similar ingredient claims, but not all of them are built on equally strong delivery architecture. When you evaluate bakuchiol systems through that lens, you move from trend participation to formulation strategy.
EXO DDS Bakuchiol FAQs
Start where your claims and base can realistically support performance. Many teams prototype within the supplier’s recommended range for the ingredient system, then confirm stability, sensory, and tolerability in the finished formula before finalizing marketing language.
For vesicle-based systems, a common best practice is late-stage addition under controlled mixing to limit mechanical stress and unnecessary heat exposure. Add where your batch conditions support uniform dispersion without destabilizing viscosity or appearance.
Delivery systems can be more sensitive to certain environments than free oils. Use extra caution with higher alcohol levels and harsher detergents/surfactants, and verify by stability testing (appearance, viscosity, odor, and performance proxies) in your exact base.
Bakuchiol delivery systems are commonly used in leave-on serums and creams where consistent skin contact time supports benefit perception. Masks and ampoules can also work well if the base avoids conditions that compromise the delivery structure and remains stable over shelf life.
Stronger claims focus on visible outcomes and user experience without overreaching: “helps improve the look of fine lines,” “supports smoother-looking skin,” and “designed for better routine comfort.” Final wording should be supported by your finished-formula testing and region-appropriate claim guidance.
Ready to evaluate EXO DDS Bakuchiol?
Take the next step from insight to action. Review the data, download the brochure, and explore where EXO DDS Bakuchiol may fit into your next retinol-alternative concept—especially when delivery, stability, and tolerability are part of the performance brief.
Forward this article to your Deveraux account managerResources
- Bluemke, A., Ring, A. P., Immeyer, J., Hoff, A., Eisenberg, T., Gerwat, W., et al. (2022).
Multidirectional activity of bakuchiol against cellular mechanisms of facial ageing—Experimental evidence for a holistic treatment approach.
International Journal of Cosmetic Science, 44, 377–393.
https://pmc.ncbi.nlm.nih.gov/articles/PMC9328396/ - Dhaliwal, S., Rybak, I., Ellis, S. R., Notay, M., Trivedi, M., & Libby, R. (2019).
Prospective, randomized, double-blind assessment of topical bakuchiol and retinol for facial photoageing.
British Journal of Dermatology.
https://pubmed.ncbi.nlm.nih.gov/29947134/ - Global Cosmetic Industry. (2026, January 28).
Retinol reimagined: The brands and ingredients gaining market momentum.
https://www.gcimagazine.com/brands-products/skin-care/news/22954993/retinol-reimagined-the-brands-and-ingredients-gaining-market-momentum - Hadiwidjaja, M., et al. (2024).
Comparative efficacy of bakuchiol oil and encapsulated bakuchiol cream on facial skin quality: A 28-day pilot study.
Journal of Pharmacy & Pharmacognosy Research, 12(3), 477–486.
https://jppres.com/jppres/pdf/vol12/jppres23.1812_12.3.477.pdf - Kuśmierska, M., Kuśmierski, J., Martyka, A., & Ujma, P. (2024).
Redefining skin health: The potential of plant-based bakuchiol as a sustainable substitute for retinoids.
Our Dermatology Online, 15(4), 345–348.
https://www.odermatol.com/odermatology/20244/3.Redefining-Ku%C5%9BmierskaM.pdf - Deveraux Specialties. (n.d.).
EXO DDS Bakuchiol (product page).
https://www.deverauxspecialties.com/product/exo-dds-bakuchiol/
Citation note
These resources were selected to support the blog’s argument from multiple angles: (1) a current industry-market framing of retinol alternatives
(Global Cosmetic Industry), (2) a peer-reviewed clinical comparison contextualizing bakuchiol’s visible outcomes and tolerability relative to retinol
(Dhaliwal et al., 2019), (3) open-access mechanistic/anti-aging evidence supporting bakuchiol’s activity across aging-related pathways
(Bluemke et al., 2022), (4) formulation-relevant evidence that encapsulation can change practical performance in a cream format
(Hadiwidjaja et al., 2024), and (5) a recent review-level discussion of bakuchiol’s positioning as a retinoid alternative
(Kuśmierska et al., 2024). The Deveraux product page is included to document ingredient-system specifics used for product-related statements;
claims should ultimately be validated in the finished formula and aligned to region-appropriate substantiation requirements.








