Executive Summary
EGF can underperform for one predictable reason: it’s easy to damage the system before it ever reaches skin. This article breaks down the five batch mistakes that quietly erode results—adding too hot, applying post-add high shear, building ethanol-heavy bases, creating detergent-like environments, and introducing the active at the wrong phase or timing in emulsions. You’ll leave with a practical, bench-ready checklist for protecting an encapsulated EGF (Oligopeptide-1) delivery system, including conservative processing targets (cool-down addition, controlled mixing) and compatibility guardrails (limit ethanol, minimize high-detergency solubilization, late-stage aqueous addition for O/W).
If EGF isn’t delivering in your finished product, the problem is often not the ingredient—it’s the batch. Growth factors are among the easiest “premium actives” to undermine through ordinary formulation choices: the wrong addition temperature, too much shear, solvent-heavy systems, detergent-like environments, or simply adding at the wrong time in an emulsion.
That’s why EGF can feel inconsistent across otherwise similar formulas. One team builds an elegant serum and sees strong consumer feedback. Another team uses a comparable dosage and ends up with a product that looks stable, passes basic QC, and still underwhelms in use. The difference is rarely luck. It’s usually process discipline and compatibility design.
This article is a practical prevention guide for brand and R&D teams working with EGF (Oligopeptide-1), especially when using encapsulated delivery systems. You’ll get a clear set of guardrails—temperature, shear, solvent limits, and phase/timing rules—plus a checklist you can apply during development to protect performance from bench to shelf.
Why EGF “fails quietly” in finished products
EGF has two constraints that matter in real formulation work. First, it’s sensitive. Proteins and peptides can lose functional integrity when exposed to stressors that many cosmetic actives tolerate—thermal stress, prolonged processing, aggressive mixing, or harsh chemical environments. That means a formula can look fine visually while the biology behind the claim has been reduced.
Second, EGF is delivery-limited in topical systems. Skin is designed to keep large, delicate biomolecules out. That’s why delivery strategies—encapsulation, vesicles, and other carrier technologies—are discussed so often in technical literature and why “EGF on the label” doesn’t automatically translate into “EGF where it needs to act.”
This is the bridge between technical reality and commercial credibility. Brand teams want the story: renewal, visible improvement, premium efficacy. R&D teams want proof of control: how you protected the active during manufacturing, how you avoided known incompatibilities, and how the final system supports availability. When those pieces are documented, EGF becomes a defendable engineering decision, not a fragile hope.
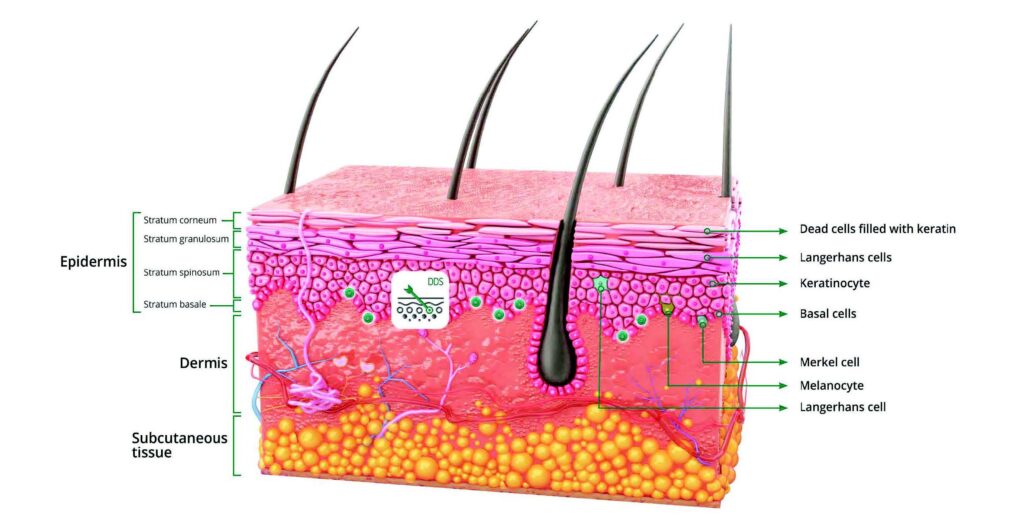
Mistake 1: Adding EGF too hot (and assuming the carrier makes it heat-proof)
Heat is one of the fastest ways to reduce performance—and one of the easiest to control once you treat it as a design variable. Many cosmetic processes require heated phases for emulsification, structuring, or polymer hydration. The mistake is allowing a growth-factor system into that hot zone.
Encapsulated systems can create a false sense of security. It’s easy to assume a vesicle or delivery carrier “solves” sensitivity. In reality, a carrier may protect against some forms of degradation or interaction, but heat is still heat. If a payload is heat-sensitive, the carrier isn’t a shield against thermal stress.
What to do instead
Treat EGF addition like you would treat other heat-sensitive materials: late-stage, controlled temperature, and minimal exposure time.
Build your base at process temperature.
Cool down into a defined window.
Add the EGF system under gentle mixing during cool-down, not during a hot hold.
A practical addition target is ≤ 40 °C at the point of addition. If your manufacturing reality demands hotter conditions, treat that as a process constraint and redesign the sequence rather than forcing the active to survive conditions it wasn’t meant to handle.
What this buys you
You remove one of the highest-risk variables for performance inconsistency. You also gain a clean line you can use in internal review: “Active system added under controlled cool-down conditions to reduce thermal stress and preserve functional integrity.”
Mistake 2: Using high shear after the active system is already in
High shear is useful. It can tighten droplet size distribution, reduce visible instability, and refine aesthetics. The problem is using it at the wrong time—after a fragile active system has been introduced—because the step is habit rather than necessity.
In growth-factor work, shear should be intentional. If you need homogenization to build the emulsion structure, do it before the active system enters the batch. Once you introduce an encapsulated payload, the goal shifts from “optimize structure aggressively” to “disperse uniformly with minimal stress.”
What to do instead
Split the process into two phases: structure-building and active protection.
- Structure-building phase: Use your preferred shear strategy to build a stable base.
- Protection phase: Cool down and introduce EGF late under controlled, lower-stress mixing.
- Finishing phase: Mix only as long as needed for uniform dispersion.
A practical limit used for encapsulated systems is a maximum homogenization ceiling of 20,000 rpm. Just as important as rpm is exposure: long mixing windows can be as damaging as high intensity.
What this buys you
Consistency. If performance varies by batch, you now have a clear audit point: shear intensity and duration after active addition. That makes troubleshooting faster and reduces “mystery variability” that’s hard to explain to stakeholders.
Mistake 3: Designing an ethanol-heavy system that destabilizes vesicles
Ethanol is common in modern sensory design. It can reduce tack, improve dry-down, sharpen slip, and support certain preservation strategies. But solvent-heavy systems can undermine delivery technologies that rely on lipid structures.
This is where performance can quietly disappear. A formula can feel elegant and still compromise the integrity of a vesicle-based system. It’s also easy for alcohol content to climb unintentionally—through solvent carriers in botanical extracts, fragrance solubilization systems, or incremental additions across multiple inputs.
A practical compatibility rule for many liposome-like delivery systems is to avoid ethanol above ~15%. Above that level, vesicle integrity may be at risk.
What to do instead
Decide what is non-negotiable in the brief.
- If EGF performance is the hero, design sensory and preservation without pushing ethanol into the zone where vesicles are likely to destabilize.
- If the product concept requires high alcohol, consider whether a vesicle-based EGF system is the right tool for that format—or whether a different active strategy is more robust.
A useful development habit is to track total ethanol equivalents during formulation the same way you track total fragrance load: as a real parameter that influences performance, not just aesthetics.
What this buys you
A direct reduction in risk. You avoid a compatibility failure that can look like “EGF doesn’t work” when the real issue is that the base environment compromised the delivery system.
Mistake 4: Creating “hidden detergency” that breaks delivery systems
Not all destabilization looks dramatic. Sometimes the system fails because the formula environment behaves like a detergent bath, even though the product isn’t a cleanser.
This shows up in modern clear formats and “water-light” serums where clarity is achieved through heavy solubilization. It can also show up when teams add solubilizers late to fix haze, carry fragrance, or force transparency. The formula may remain elegant and stable while delivery systems become less intact.
A practical compatibility principle is simple: high detergency environments can break vesicle systems. If the formula behaves like it wants to solubilize lipids, it may also disrupt the lipid architecture of your delivery carrier.
Where hidden detergency commonly appears
- Micellar-adjacent serums
- Clear gels with aggressive solubilizer stacks
- Systems using surfactants for sensorial slip rather than structure
- Late-stage clarity “fixes” that increase solubilizer load right when the active system is most vulnerable
What to do instead
Treat surfactant/solubilizer load as a compatibility budget.
- Choose emulsifier and solubilizer systems that meet the sensory target without pushing the formula into detergent-like behavior.
- Reduce the need for solubilizer stacking by designing clarity intentionally from the start.
- Avoid last-minute adjustments that add detergency after the delivery system is already in.
What this buys you
Performance that survives the final “polish.” Many formulas lose the active story during small late-stage modifications. If you control detergency, you protect the delivery system through the part of development where shortcuts tend to creep in.
Mistake 5: Adding at the wrong time or in the wrong phase in emulsions
Emulsions are where premium anti-aging concepts often live—and where growth-factor systems are most likely to be stressed. Not because emulsions are incompatible, but because the process includes multiple stress points: heat, emulsification energy, shear, and surfactant architecture.
For oil-in-water (O/W) systems, the most defensible and repeatable approach is typically to introduce vesicle-based actives into the aqueous phase and/or at the last stage of manufacture. The goal is simple: keep the delivery system out of the harshest part of processing and avoid making it participate in the structural formation of the emulsion.
What to do instead
Build the emulsion first. Let structure form. Then add the EGF delivery system during cool-down under controlled mixing.
- Final phase addition (cool-down)
- Gentle mixing
- Minimal exposure to hot holds or aggressive shear
- Stable base first, active system second
What this buys you
A manufacturing story you can defend. In technical review, the question is rarely “Did you add EGF?” It’s “How did you keep it intact and functional in the finished system?” Phase and timing control is the cleanest answer.
The practical checklist: keep EGF working from lab to shelf
If you want the bench-ready version, start here. This is the checklist that prevents most quiet failures.
Process guardrails
- Add EGF systems during cool-down; target ≤ 40 °C at addition
- Avoid aggressive post-addition stress; keep shear conservative after the active is present
- If homogenization is required, keep it controlled (a practical ceiling often used is ≤ 20,000 rpm)
- For O/W emulsions: introduce in the aqueous phase and/or last stage of manufacture
Formulation guardrails
- Keep ethanol conservative; avoid > ~15% ethanol in vesicle-based systems
- Avoid high-detergency environments; minimize detergent-like surfactant/solubilizer load
- Confirm pH compatibility within the supplier’s recommended window and within your finished product goals
These aren’t restrictions. They’re design inputs. When a brief starts with them, development moves faster, performance is easier to defend, and the final product is less dependent on “perfect handling.”
Why encapsulated EGF is used in the first place
Once you accept that EGF is sensitive and delivery-limited, delivery systems stop looking like a marketing flourish and start looking like a practical response to known constraints.
A well-designed encapsulation approach is trying to solve three problems at once:
- Protection: reducing exposure to destabilizers that can degrade or inactivate the peptide.
- Availability: improving the chance that the payload remains available where it can influence cosmetic endpoints.
- Repeatability: narrowing the window of easy mistakes so performance doesn’t drift batch-to-batch.
In supplier technical materials for encapsulated EGF systems, you will often see emphasis on gradual delivery, improved bioavailability, and protection against degradation—because those are the real bottlenecks in topical growth-factor work. You may also see practical benefits like reduced appearance drift (for example, minimizing color change), which matters for premium products where consumer trust is tied to consistency.
The key to keeping this credible is the framing: encapsulation is not magic. It’s engineering. It improves your odds—if the formula and process are designed for compatibility.
How INdermal’s encapsulated EGF fits into real product development
In practical formulation terms, the encapsulated EGF system discussed here is designed to help teams manage the two big constraints: sensitivity and delivery. It also comes with clear “how to not break it” guidance that makes it easier for R&D teams to build stable processes.
Where it fits best depends on your format and your tolerance for compatibility risk:
- Serums and gels can be ideal when you need controlled temperature at addition and want to keep alcohol and detergency low.
- O/W emulsions are viable when you can add late-stage, control post-addition shear, and avoid solvent-heavy or detergent-like environments.
- Premium anti-aging systems benefit most when the active story is central enough to justify process discipline.
If you need performance-supporting evidence for internal alignment, INdermal also provides an ex vivo dataset using a human organotypic skin explant model comparing an encapsulated EGF gel to a free EGF gel at the same concentration, assessing collagen- and elastin-related outcomes via histological methods. It’s not a clinical trial, but it is a structured comparative approach that can help inform development decisions and support technical positioning during customer conversations.
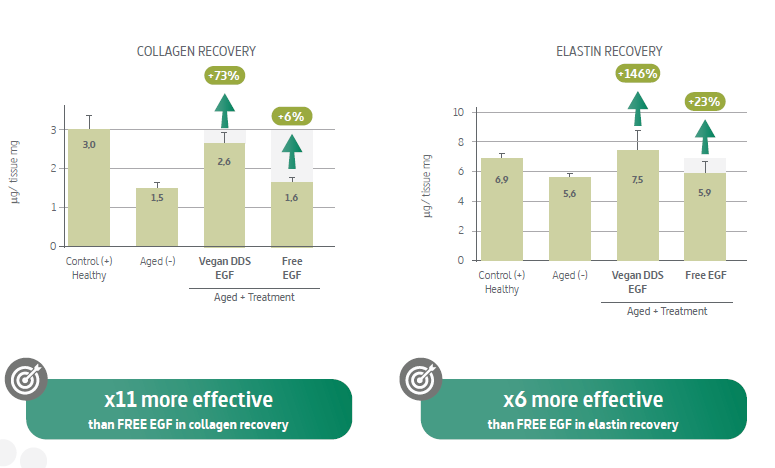
What to do next if you’re evaluating EGF for a new brief
If your team is considering EGF for a launch, don’t start with claims language. Start with compatibility.
- Choose the base format based on solvent and surfactant requirements.
- Lock the manufacturing sequence early (cool-down addition, low-stress finishing).
- Set a compatibility budget for ethanol and detergency rather than discovering the problem late.
- Validate reproducibility, not just appearance stability—because the quiet failures are often functional.
When those pieces are in place, EGF becomes manageable. It stops being a fragile variable and becomes what it should be: a premium active with a process designed to keep it effective.
EGF Formulation FAQs
For many development programs, a common starting range is 1–3% in leave-on serums, gels, and O/W emulsions, then adjust based on stability, sensorial impact, and your finished-formula testing plan. If you’re targeting a “hero active” position, evaluate higher levels only after confirming compatibility and batch reproducibility in your specific base.
Add during cool-down and keep the addition step ≤40 °C. In emulsions, introduce the system in the aqueous phase and/or at the last stage of manufacture, then mix only until uniform. This reduces exposure to the highest thermal and mechanical stress points in the batch.
As a conservative rule, cap post-add mixing at a maximum homogenization of 20,000 rpm, and avoid long high-shear windows after addition. A reliable approach is to do any necessary high-shear work earlier (base formation), then switch to gentle, short-duration mixing after adding the encapsulated EGF to maintain delivery-system integrity.
Be cautious with solvent-heavy designs. A practical compatibility guardrail is to keep ethanol <15%, since higher levels can damage liposome-like delivery systems. Also watch “hidden ethanol” from extracts and fragrance carriers. If the concept depends on high alcohol for sensorial reasons, validate vesicle integrity and performance in stability before committing.
The most common risk is a detergent-like environment from high surfactant or solubilizer load (clear “micellar-adjacent” serums, heavy fragrance solubilization, or late-stage clarity fixes). Too much detergency can break liposomes. If you need clarity and fragrance, treat solubilizer level as a compatibility budget and confirm stability, viscosity, and appearance over time in the finished base.
Ready to evaluate Vegan DDS EGF?
Take the next step from insight to action. Review the technical brochure and explore where Vegan DDS EGF may fit into your next leave-on serum, gel, or O/W emulsion—while keeping batch handling and compatibility guardrails in view.
Forward this article to your Deveraux account managerResources
- Eskens, O., et al. (2021). Challenges and effective routes for formulating and delivery of epidermal growth factors in skin care.
International Journal of Cosmetic Science.
https://pubmed.ncbi.nlm.nih.gov/33354795/ - Shin, S. H., et al. (2023). The use of epidermal growth factor in dermatological practice.
Dermatology Practical & Conceptual.
https://pmc.ncbi.nlm.nih.gov/articles/PMC10333026/ - Andra, V. V. S. N. L., et al. (2022). A comprehensive review on novel liposomal methodologies, preparation, characterization, and applications.
Journal of Drug Delivery Science and Technology.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8790012/ - Pilch, E., et al. (2018). Liposomes with an ethanol fraction as an application for drug delivery.
International Journal of Nanomedicine.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6320757/ - Nsairat, H., et al. (2022). Liposomes: Structure, composition, types, and clinical applications.
Heliyon.
https://www.sciencedirect.com/science/article/pii/S240584402200682X
- INdermal / Nanovex Biotechnologies. (n.d.). Vegan DDS-EGF_01 Brochure v05. PDF provided by Deveraux Specialties.
- INdermal / Nanovex Biotechnologies. (n.d.). Vegan DDS-EGF – Ex vivo efficacy study: Firming activity. PDF provided by Deveraux Specialties.
Citation note:
These sources were selected to support the article’s core technical argument without relying on promotional claims.
The growth-factor review literature supports the key constraints formulators face (delivery limits, stability, and interactions in cosmetic systems).
The liposome/vesicle references provide mechanism-level context for why solvent composition and detergent-like environments can destabilize
vesicle-based delivery systems. Supplier documents were used only for specific, actionable processing and compatibility guardrails, plus
product-specific facts and the existence/structure of the ex vivo comparative dataset.








